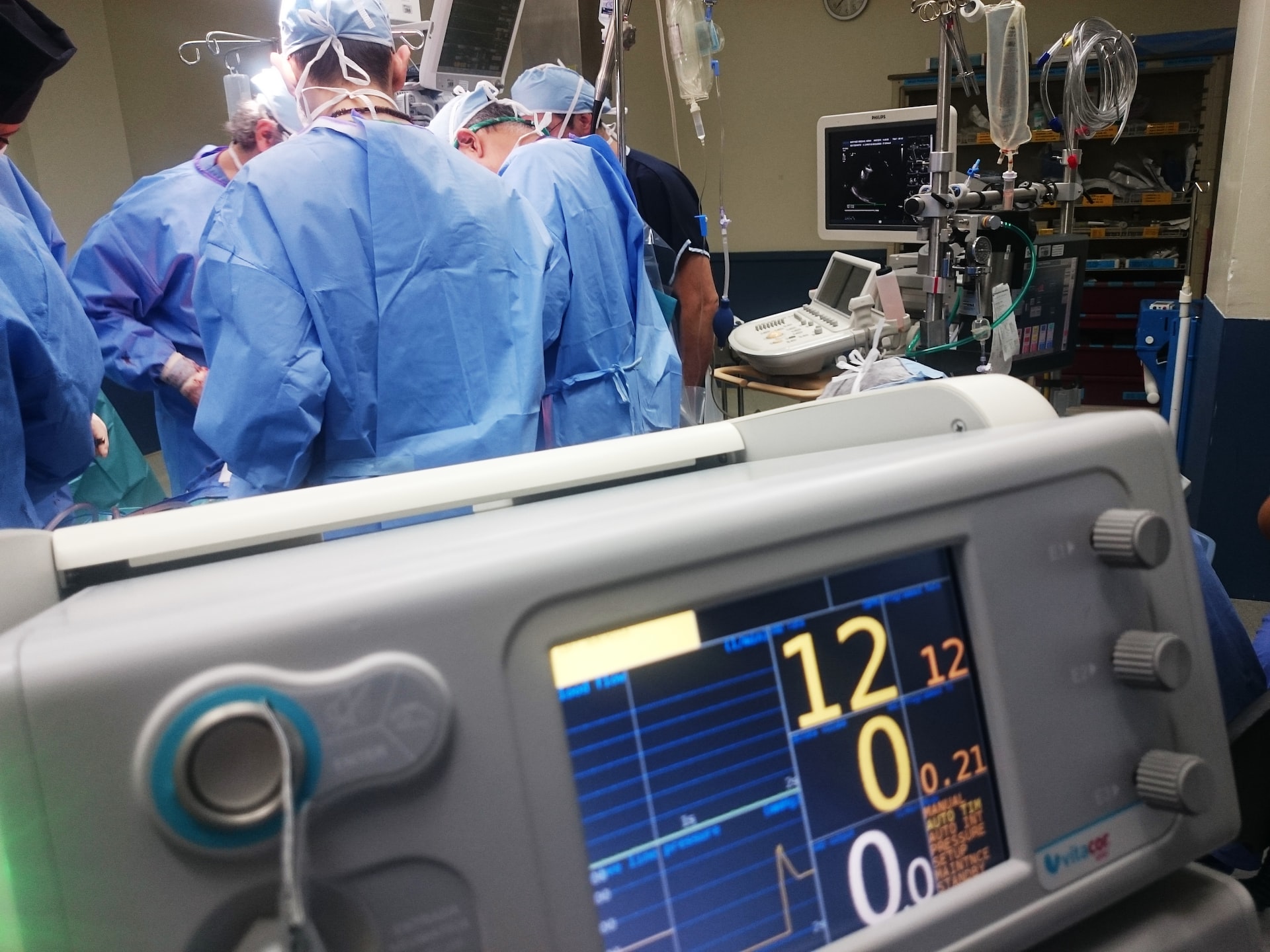
The role of a biomedical engineer is pivotal in the smooth functioning of hospital equipment and departments.
A biomedical engineer takes diligent care for the smooth functioning of medical devices and also manages the overall structuring of a hospital like various departmental planning, involving in designing the floor plan of hospital, procurement of higher end equipment followed by preventive maintenance of the same.
To explain in brief, the responsibilities of a biomedical engineer begins with hospital planning, calculation of ROI (Return on investment), purchase of equipment, installation, commissioning, training, creating the master asset list of the devices in the entire hospital, QA(Quality Assurance) for radiology devices and managing the gas plants etc. Once the device is in use, it has to undergo periodic action plans and management of maintenance contracts along with calibration. Finally all the above processes need to be stored and documented in the database for future reference
Starts from :
- Planning: In this phase, a biomedical engineer needs to have a thorough plan of devices ranging from lower risk to high risk which will suit the hospital depending on the geographical area and other aspects.
- Pre-purchase Evaluation & Negotiation in Procurement: This is the most time consuming and brainstorming part among all the processes, wherein a complete study needs to be done before procuring a device in hospital, followed by a fair and ethical negotiation, later comes the purchase and related paper works.
- Commissioning and Installation Post Procuring the Device: The purchased equipment to be matched with the purchase order, then to be installed and commissioned. This is where service engineers require full support from biomedical engineers to run the installation work and also to check on the device’s performance and operation.
- Training: In this phase, biomedical engineers give training to healthcare workers associated with the device to teach them the complete functioning and application of the device. In regard with the training sessions conducted, engineers need to get a document signed stating the successful completion of the training. Refresher training is also conducted under this category, where training is given periodically to keep the healthcare workers updated on new device upgradations.
- Breakdown Management: Under breakdown management the Six Sigma methodology- DMAIC- (Define, Measure, Analyze, Improve and Control) has a major role, which help the engineers to ease their work and move in a streamlined flow. This includes various categories like management of maintenance contracts, analysis of equipment failure, assessment of repair cost, scheduling of repair/PM visits, monitoring and documentation
- Incoming Inspection of Equipment : This includes various audits like NABL, NABH and other international accreditation like JCI and ACHSI. The reports and documents related to each device needs to be submitted to the auditors.
- Equipment Risk Management: The other sole responsibility of a biomedical engineer is risk management, where the devices will be provided with risk classes while testing, followed by a computerized medical management system, which is a perfect tool for tracking device inventory and maintenance history. This in turn reduces the preventive maintenance requirement and internal device surveillance along with changes in standards.
- Documentation of all Condemnation-if not repaired or out of use: The devices which cannot be used or repaired should be labelled as condemned devices, such category devices need to be listed and documented.
The responsibility of a biomedical engineer is not just limited to device handling, but also to ensure that the entire environment where the device fits into is well managed and maintained without fail. The proper and timely practice of these steps will offer standard healthcare services to the public. Hope this article on the roles and responsibilities of a biomedical engineer in a hospital is insightful.